Idarucizumab reversal6/29/2023 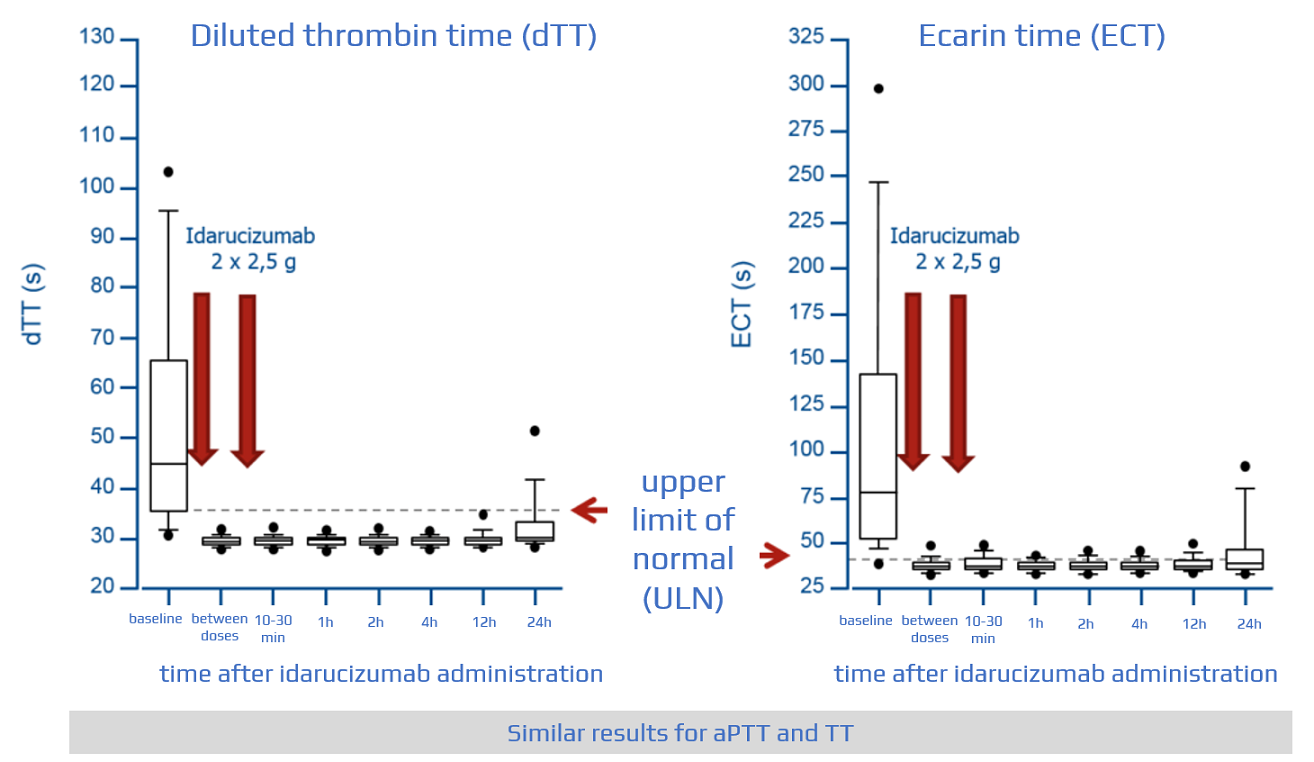
However, re-occurrence of dabigatran effect was noted later in four patients with creatinine clearance less than 30 ml min −1, and one patient with persistent bleeding required retreatment with idarucizumab. After idarucizumab administration, normal coagulation parameters were confirmed in 10/11. Among cases with laboratory data available, baseline coagulation tests were prolonged in 12/13 cases with bleeding or emergency surgery. Five patients died due to cardiogenic, haemorrhagic or septic shock, intracranial bleeding or multiple organ failure. During surgery, no excessive bleeding was reported. The indications for idarucizumab use were: emergency surgery (4/17), severe bleeding (11/17 seven with intracranial bleeding) and ischaemic stroke (2/17). One patient was treated with the antidote twice with an interval of 2 months between treatments. The decision to reverse dabigatran with idarucizumab was made by the treating clinicians, as was the assessment of clinical outcomes and blood sampling/monitoring (activated partial thromboplastin time, thrombin time and diluted thrombin time) before and after use. 
We analysed consecutive cases treated with idarucizumab in Slovenia from January to October 2016. To evaluate efficacy and safety of the clinical use of idarucizumab after its availability in Slovenia. 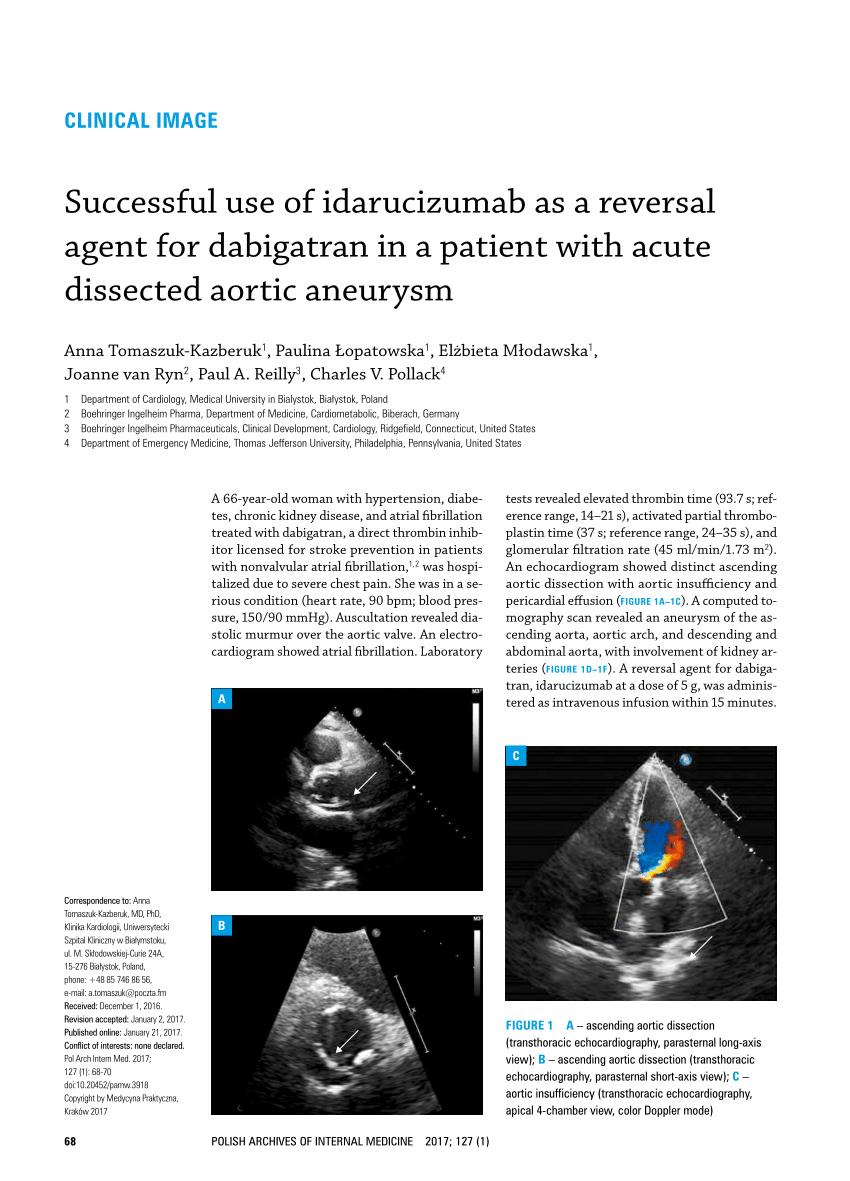
Real-life experience with idarucizumab, which reverses the anticoagulant effect of dabigatran, is currently limited.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
